|
By: Daniel Wright
Illustration: Declan Wrede
FDA and Patent Timeline Mismatch
In its mission to protect citizens from faulty or fraudulent products, the Food and Drug Administration (FDA) requires you to submit substantial proof of both efficacy and safety of your new pharmaceutical or medical device before it can be legally sold in the United States. Collecting these data in clinical trials and the subsequent review process takes years, sometimes over a decade, and if there’s a patent involved, it’ll likely issue well before the FDA gives you the green light. This can seem more than a bit unfair since that patent term is supposed to provide the economic exclusivity needed to recuperate the fortune expended on drug development. Rest assured, the FDA is aware of this counterproductive interaction and offers a number of compensatory options. First, though, let’s take a look at the patent prosecution and FDA approval timelines for context.
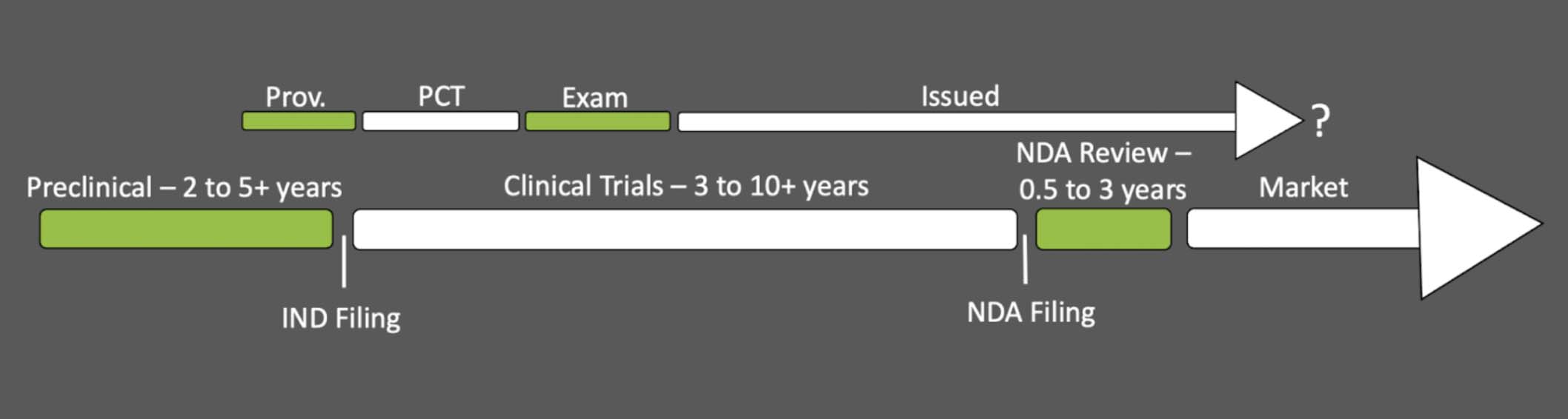
MATCHING PACE: Patent Prosecution Timeline
The patent prosecution timeline has a few options depending on your target geographic market and whether you expedite your application, but shown here is the common path. Most applicants begin with a provisional application that yields an international patent application filing, usually under the Patent Cooperation Treaty (PCT), enabling the application’s entry into foreign jurisdictions. The duration of examination before each local patent office can vary dramatically, but in the U.S., this can take around one to three or more years. Following approval, your issued U.S. patent will remain enforceable for the patent’s term of twenty years starting from your first non-provisional filing date (as long as you keep paying the maintenance fees). Therefore, from start to finish, you’re looking at about four or more years to acquire your issued patent which will last for about sixteen years afterwards (note: the provisional patent application year is not counted in your 20 year patent term).
MATCHING PACE: Drug Discovery and Regulatory Timeline
Now, let’s review the other half of this discussion. The patent prosecution timeline can seem a mere blink of an eye in comparison to the duration that drug discovery and the regulatory process demand.
Preclinical research. The process begins in preclinical research. Here, chemists and biologists will decide on their biological target for the disease indication (e.g., which protein to inhibit to treat what illness), and design molecules to test for viable activity (e.g., does it actually inhibit the desired protein). Promising compounds will then be pushed into cell lines and animal models to probe for the drug’s in vivo efficacy, bioavailability, and toxicity.
Investigational New Drug (IND) Application. With promising preclinical results in hand, you can then approach the FDA with an Investigational New Drug (IND) Application. In this document, you present all your evidence that suggests efficacy and safety in humans along with the parameters of your proposed clinical trials. The FDA will review it within thirty days for any unreasonable risk, demanding further experiments if needed, but, in bestowing approval, will authorize you to commence your clinical trials.
Clinical trials will likely be the longest period of this process because of the sheer scope of the undertaking. The mobilization of a sufficient patient population to explore short and long term effects in rigorous studies is not something that can be performed over a weekend, and for most, this operation will take about six to seven years, sometimes longer. New Drug Application (NDA). Ultimately, you will compile your (hopefully) successful clinical trial results along with relevant preclinical insights into a New Drug Application (NDA) that demonstrates the safety and efficacy of your new pharmaceutical towards one or more specified disease indications. The FDA has up to three years to return a final decision. If they find any aspect lacking, they may require additional studies to be performed; otherwise, with approval of the NDA comes your ability to finally sell your new pharmaceutical, perhaps only ten to fifteen years after you started. Note: Medical devices go through a very similar process, and if your drug is a generic of a compound previously approved by the FDA you can likely skip the clinical trials (we won’t be discussing those scenarios here). Patent Prosecution and FDA Timelines Side-by-Side
Having now explored both the patent prosecution and FDA timelines, let’s place them side by side. The exact alignment will depend upon when you chose to file your patent, but for many, the provisional application will be filed sometime near the IND filing due to the somewhat public nature of clinical trials and the concurrent demand for more open advertising and public relations. Accordingly, we end up with a dual timeline that looks something like this:
Very likely, you’ll acquire your issued patent sometime during clinical studies, well before you can enter the market. In the worst case scenario, your issued patent may have less than 5 years of life left when you can finally enter the market with your new drug. Let’s take a look at what the FDA offers as compensation for this less-than-ideal situation.
Options for Maximizing Your Exclusivity Window
In return for the extra regulatory hurdle and the potential lost patent protection time, the FDA offers the following three perks in certain conditions:
Patent Term Extension (PTE) means exactly what the words say: bonus term for your patent. Upon approval and at your timely request, the FDA will instruct the United States Patent and Trademark Office to append all of the time spent in NDA review and half the time between the IND and NDA filings to a single relevant patent. Up to a combined total of five years can be added, and the remaining lifetime of the chosen patent, after the addition, cannot exceed fourteen years. Furthermore, only the relevant claims enjoy the extension, so if your selected patent claims three discrete compounds separately, only the one towards the approved drug gets extended. The remaining two perks function independently of any related patents and instead only affect the FDA’s operations in your favor. New Chemical Entity (NCE) Exclusivity is an award to new “active moieties” of a five-year period in which the FDA will not accept any competing applications (usually an application for a generic of your drug). This means that, even if you have no patent on the drug, your successful FDA approval grants you a functionally similar limited monopoly on the market while the regulatory agency refuses to even initiate the process with competitors. Note that the FDA considers the “active moiety” of a drug to be the biologically active chemical entity regardless of any formulation, salt, or prodrug. If you’re working with any of the latter, you’ll be interested in what’s next. New Clinical Investigation Exclusivity is a three-year period in which the FDA will not approve competing applications. New Clinical Investigation Exclusivity is awarded to successful cases that involve a previously approved active moiety but had required new clinical studies. Applications that repurpose an old drug in a new formulation, prodrug, dosage, or towards a new disease indication can all qualify, but keep in mind that this perk is necessarily narrower than NCE Exclusivity. Assuming there’s no broader protection for your product, competitors could avoid your New Clinical Investigation Exclusivity by remaining with an older, unprotected formulation or marketing their generic only towards previously approved diseases. Start synchronizing your timelines…first steps to restful nights
The FDA certainly adds a whole other layer of complexity to the question (and we did not discuss here more niche perks such as those to pediatric or orphan drugs nor the nuanced tactics competitors can try to circumvent the various FDA exclusivities), but it’s comforting nonetheless that the agency is aware of the barrier its efforts present to the patent system and its motivating economic incentives. Certainly, you should discuss your filing strategy with your practitioner before committing to any plan, but you can sleep soundly knowing that some options remain even if your patent issues (or worse yet, is near expiration) well before your drug is on the market.
1 Comment
By: Ashley Sloat
Women's History Month: Meet Mary Kies
As part of our contribution to celebrating Women's History Month, we're introducing you to Mary Kies, America's First female patent holder!
On May 15, 1809, Mary became the first woman to be awarded a US patent. This was a full 19 years after the passage of the 1790 Patent Act that allowed "any person or persons" to petition the US government for protection of their original methods and designs. The Patent Act technically opened the door for both men and women to protect their inventions, but at the time, in many states, women couldn't own property independent of their husbands or fathers. As a result, many women never tried, but Kies was an absolute pioneer in breaking that trend. A Fashionable Solution to an Economic Problem Her patent was a method for weaving straw and silk. The Napoleonic Wars between France and Britain had resulted in a trade embargo on British goods in 1807. This embargo was economically devastating and forced the early American fashion industry to be less dependent on imports. New England women turned to straw hat making as one DIY solution. Kies' approach involved weaving silk into the straw, creating a more aesthetically pleasing appearance and kicking off an early American fashion fad. James Madison signed this patent into law and reportedly, Dolley Madison wrote Kies a letter, congratulating her on her contribution to helping women in history. Still a Gap 232 Years Later We've come a long way since Mary's time, but even now, less than 22% of U.S. patents have at least one woman inventor named and among the entire US inventor demo, women only account for 12% of inventor-patentees. Be sure to check out our Innovation is for Everyone post to learn more about the USPTO's Expanding Innovation Initiative that includes a free crash-course on the fundamentals of patent prosecution, as well as opportunities for mentoring, and access to community groups.
By: Josh Sloat
S2 Episode 1: Software Patents
Protecting your digital innovations
We're officially kicking off the second season of the Patently Strategic Podcast after taking a brief post-holiday break and in this month's episode, our experts tackle software patents. As Silicon Valley engineer and investor Marc Andreessen famously said, “Software is eating the world”. It is central to so much of today’s innovation and the growth potential is off the charts. Despite rate hikes rattling the market a bit, Microsoft, Apple, Google, Amazon, and Tesla – these 5 companies, all with software at their core are worth a quarter of the S&P’s entire market cap. It’s possible that Meta Platforms, formerly known as Facebook, could eventually be the sixth such company worth over $1 trillion. In our microcosm of focus, these eye-popping stats are no exception. The AI powered market will grow to $180 billion by 2025. By the end of this year, the Global Edge Computing market will reach $6.72 billion and the global AR and VR market will exceed $209 billion. By 2030, there will be 50 billion IoT devices in use worldwide. That’s 6 devices each for every man, woman and child on this planet – and all of them are powered by software. The stakes are high, the value is soaring, and the importance of quality patenting has never been more strategically vital. In this month’s episode, Dr. Ashley Sloat, President and Director of Patent Strategy here at Aurora leads a discussion along with our all star patent panel, digging into:
The group also concludes with an insightful brainstorming session around two problems that plague this space. The first is around a new patent type that would help overcome many of the current prosecution and court problems faced by software patents. The second, pulling from adjacent IP law, is a mechanism for small inventors to receive compensation for using their patents, similar to how music copyright royalties are handled. Ashley is joined today by our always exceptional group of IP experts including:
Availability
Patently Strategic is available on all major podcasting directories, including Apple Podcasts, Spotify, and Google Podcasts. We're also available on 12 other directories including Stitcher, iHeart Radio, and TuneIn, so you should be able to find us wherever you listen to podcasts.
Topic and guest participant requests
If you’re an agent or attorney and would like to be part of the discussion or an inventor with a topic you’d like to hear discussed, please reach out.
Resources
By:Josh Sloat
Episode 9: Patent Anatomy
What's in a patent?
What’s actually in a patent? What can you learn from reading one? Where do you even find them? For answers to these critical questions and more, I sat down with Patently Strategic regulars – David Cohen, Principal at Cohen Sciences and Ashley Sloat, President and Director of Patent Strategy here at Aurora. In our discussion, we break down the key anatomy, cut through the terminology, and provide tips on how you can use this information for your competitive advantage. Patenting can be such an essential part of early startup years, but it’s a very complex domain, with loads of special vocabulary, and all of that can make it seem less accessible than it was really designed to be. In putting the questions together for the Q&A with Ashley and David, I quickly realized that, had I had the answers and this context much sooner in my own journey into this world, a lot of things would have made a lot more sense much earlier. This is a guided tour, intended to reduce this barrier of entry, unlock for you one of the greatest libraries in the world, and help set you up with clear eyes going into your own patent journey. The tour is done through the anatomical lens of the most intrinsic and tangible piece of the process – the patent application. It’s through the study of a patent's structure and parts, that you’ll quickly begin to see more clearly how the broader world of patenting works! Availability
Patently Strategic is available on all major podcasting directories, including Apple Podcasts, Spotify, and Google Podcasts. We're also available on 12 other directories including Stitcher, iHeart Radio, and TuneIn, so you should be able to find us wherever you listen to podcasts.
Topic and guest participant requests
If you’re an agent or attorney and would like to be part of the discussion or an inventor with a topic you’d like to hear discussed, please reach out.
Resources
Throughout the conversation, we mentioned a handful of useful sites for searching patents:
We're also providing computer-generated transcripts for improved accessibility and additional reference opportunities.
By: Josh Sloat
Episode 8: Inventorship
Who should be listed as an inventor for a patent?
In the US especially, patents are granted to inventors. More often than not, that’s more than one person and the ideas themselves are fluid concepts that often evolve through many handoffs from initial conception through implementation and sometimes even throughout patent prosecution, but how do we determine who all should – and is legally required to be – officially named as an inventor? In the constitutionally expressed interest of protecting inventors and the conception of their ideas, failure to include the right people can be a death sentence for a patent and grounds for invalidity. In this episode, Daniel Wright, Partnership Manager and Patent Strategist here at Aurora, will lead a deep dive into the origins of inventorship, break down who is and isn’t eligible for inclusion as an inventor, and explain how improper inventorship could result in revoked patent rights. Daniel is joined today by:
Availability
Patently Strategic is available on all major podcasting directories, including Apple Podcasts, Spotify, and Google Podcasts. We're also available on 12 other directories including Stitcher, iHeart Radio, and TuneIn, so you should be able to find us wherever you listen to podcasts.
Topic and guest participant requests
If you’re an agent or attorney and would like to be part of the discussion or an inventor with a topic you’d like to hear discussed, please reach out.
Resources
For the visual learners out there, we also like to make our presenter slides available for your reference. This particular deck includes great visuals for the guided invalidity search walkthrough.
We're also providing computer-generated transcripts for improved accessibility and additional reference opportunities.
By: Josh Sloat
Illustration: Declan Wrede
SPARK.ed Patenting Series
Aurora is partnering with Ann Arbor SPARK to deliver a three part SPARK.ed series. You've had questions around patenting and the SPARK team has been listening! We've put this series together to address the most common questions that arise from startups within the SPARK ecosystem. Our very own Ashley Sloat, Ph.D. will be the instructor for all three talks.
Part 1: Patenting in the Early Years: A coming-of-age story Part 2: Decrypting Software Patents: Key Insights for IP Success Part 3: Prenuptial Patenting
Responsible Engagement with Engineering Firms
You have your big idea and now it’s time to breathe it into existence, but you need some help with the development. In this talk, we discuss everything you need to know before and during the course of engaging with an engineering firm. We’ll cover IP ownership, assignment from engineering firm inventors back to you, and how to avoid the traps of viral IP. Time and Location: Thursday, December 9, 2021 at 11:30am - 12:30pm | Free Online Event Patent Education for You!
To help reduce the IP barrier of entry for early stage founders and startups, we regularly make our talented staff available for speaking engagements at conferences, competitions, accelerators, and incubators. We also partner with groups like Ann Arbor SPARK, XLerateHealth, and InnovatorMD to host live webinars. Please reach out if you'd like to schedule a talk or workshop for your group, and see our Events page for a sampling of past and upcoming talks.
By: Josh Sloat
Illustration: Declan Wrede
Episode 7: American Inventor Horror Story
10 Years of AIA and the PTAB
Listen in as we discuss the impact of the AIA and the PTAB with inventors gathered as part of the "Decade of Stolen Dreams" rallies, happening in front of regional USPTO offices all across the country, marking the 10 year anniversary of the passage of the America Invents Act – an event described by advocacy groups as the worst event in U.S. patent history. The inventors and entrepreneurs at the rally we attended, all from diverse backgrounds with very different stories, have one thing in common and that is their shared belief that the AIA and the PTAB – with its eye-popping 84% invalidation rate – have crippled innovators and created a Decade of Stolen Dreams, ruining the lives of countless inventors and shutting down numerous start-ups, in favor of Big Tech and multinational corporations. In this episode, we break down the AIA and PTAB through a wide array of personal perspectives from inventors, patent practitioners, and even a former USPTO patent examiner. We explore its origins, core problems, and proposed solutions. We also provide some very practical tips that inventors should consider now to help future-proof their patents, should they ever find themselves on the receiving end of an IPR. Inventors are not only the real engine of our economy, but they’re also the reason we do what we do here at Aurora. This is a complex issue, with high stakes, involving the people who matter most to our business and what the world will look like for our kids, so we can’t think of a better use of a podcast episode than to lend a voice and hopefully shine a light on the key issues and some potential solutions. I am joined today by an exceptional group of industry experts, founders, and inventors including:
Availability
Patently Strategic is available on all major podcasting directories, including Apple Podcasts, Spotify, and Google Podcasts. We're also available on 12 other directories including Stitcher, iHeart Radio, and TuneIn, so you should be able to find us wherever you listen to podcasts.
Topic and guest participant requests
If you’re an agent or attorney and would like to be part of the discussion or an inventor with a topic you’d like to hear discussed, please reach out.
Resources
If you haven’t listened to our episodes on patent searching or patent monetization, we highly recommend you also give them some ears. In this episode, we talk a fair amount about importance of searching prior art and how it’ll help if you ever find yourself facing the PTAB. It tees in wonderfully with the context and strategy bits Shelley and Louis cover in the prior episodes. Be sure to check 'em out.
We're also providing computer-generated transcripts for improved accessibility and additional reference opportunities.
By: Josh Sloat
Welcome Aboard
We could not be more thrilled to be welcoming Kristen Hansen to the Aurora family! Kristen joins us this month, starting her role as a Patent Strategy Specialist. Kristen is the perfect blend of wicked patent strategy smarts, deep technical expertise, startup-focused business acumen, and all-around great human. She exudes all of the essentials that have come to define the Aurora DNA. We love working with her and know that she will be a tremendous asset to both Aurora and our clients.
Patent Strategy. Kristen is a USPTO-registered patent practitioner with over 15 years of experience focusing on developing U.S. and international patent portfolio strategies for individual, university, and corporate clients. Many of these clients include THE tech giants and Fortune 100s in the areas of software, IoT, machine learning, consumer electronics, electronic circuits, optics, medical devices, wearable devices, virtual and augmented reality technologies, telecommunications, semiconductor devices, and business methods. Software Expertise. In a world where software is dominating the technology landscape, it's essential to have someone with deep software patenting experience in your corner, ensuring protection for your most central and valuable IP. In a prior role, Kristen focused the majority of her time managing portfolios for companies like Google, Apple, SAP and Twitter, where she drafted and prosecuted mission critical patents in the areas of AR/VR implementations, browser inventions, operating system interaction, IoT, machine learning, search algorithms and techniques, mobile and desktop app implementation, business methods, and web store algorithms. Business-driven IP. Kristen understands complex technologies, knows what moves business, and is a world class communicator. As a result, she can comfortably communicate with inventors at a detailed level and can disseminate technical details of such communications to non-technical audiences. Kristen uses her technical knowledge and legal training to develop patent portfolio strategies that are closely aligned with each client’s business objectives.
By: Josh Sloat
Episode 6: Patent Monetization
Buying and Selling, with special guest host Louis Carbonneau
In this episode, we take a deep dive into the war games filled world of patent buying and selling. We are joined today by special guest host, Louis Carbonneau, Founder & CEO of Tangible IP. Mr. Carbonneau is a recognized expert in intellectual property with close to 30 years of professional US, Canadian, and international experience in all facets of intellectual property law and business. His firm is the largest brokerage firm in the world and has transacted over 4,500 patents in the past decade. When we sat down with Louis, we hoped to be able to provide our listeners with a 101 style talk on the ins and outs of patent monetization. What we got was so much more and so very consistent with our mission of helping our audience see around corners and help their future selves. Louis begins with some excellent historical perspective and an overview of the state of the market, but really drives it all home with a plethora of forward-thinking strategies that make for more robust, defendable, assertable patents. Patents are a long game. The little things you do now can have tremendous impact over the next 15 or more years of your business journey and it’s vital that inventors and practitioners start keeping these strategies in mind, if they have any hope of crafting battle ready, commercial-grade patents. Expert Panel Louis is joined today by our always exceptional group of IP experts including:
Topics Covered
Availability
Patently Strategic is available on all major podcasting directories, including Apple Podcasts, Spotify, and Google Podcasts. We're also available on 12 other directories including Stitcher, iHeart Radio, and TuneIn, so you should be able to find us wherever you listen to podcasts.
Topic and guest participant requests
If you’re an agent or attorney and would like to be part of the discussion or an inventor with a topic you’d like to hear discussed, please reach out.
Resources
If you haven’t listened to our episode on patent searching, we highly recommend you also give it some ears. In this episode, Louis talks a lot about the importance of searching as it pertains to drafting valuable patents, how it’ll help if you get IPR’d, and also help against infringers. It tees in wonderfully with the context and strategy bits Shelley covers in the prior episode. Be sure to check it out.
We're also providing computer-generated transcripts for improved accessibility and additional reference opportunities.
By: Ashley Sloat
What you can do!
Please help our client, Starlight Cardiovascular, win a financial prize to further their development of devices for babies that are born with an extra connection in their heart and need lifesaving surgery.
Please like, share, and watch their one minute video until the end. See below and original link here. About Starlight Cardiovascular
Starlight Cardiovascular is focused on the essential mission of creating devices to treat babies born with congenital heart defects. Approximately 1.3 million babies are born each year with a CHD. Most of the tools used to treat these children were designed for adult conditions and anatomy, limiting their utility for pediatric patients. Starlight Cardiovascular is developing a portfolio of devices designed and tested specifically for pediatric CHD with the goals of improving morbidity, mortality, and access to care.
|
Ashley Sloat, Ph.D.Startups have a unique set of patent strategy needs - so let this blog be a resource to you as you embark on your patent strategy journey. Archives
April 2024
Categories |















 RSS Feed
RSS Feed